Why Cerium Oxide Excels in High-Temperature Applications —— Exploring the Thermal Stability and Redox Superiority of CeO₂
Cerium oxide (CeO₂) stands out among rare-earth materials for its exceptional thermal stability, oxygen storage capacity, and reversible redox properties, making it a critical material in high-temperature applications. This article examines how CeO₂ maintains structural integrity under extreme heat, resists sintering, and promotes catalytic activity through its Ce⁴⁺/Ce³⁺ transition. It highlights its roles in automotive catalytic converters, solid oxide fuel cells, thermal barrier coatings, and high-temperature polishing processes. The article also explores how doping and nanostructuring further enhance its performance, ensuring CeO₂ remains a material of choice for industries demanding both durability and reactivity under harsh thermal conditions.
Introduction: The Heat-Resistant Nature of Cerium Oxide
Cerium oxide (CeO₂), a rare-earth metal oxide, has become a cornerstone in modern high-temperature materials due to its extraordinary thermal stability and redox flexibility. Unlike conventional oxides that degrade or lose structure under prolonged heat, cerium oxide can maintain its crystal integrity even above 1000°C, making it ideal for demanding environments such as catalytic converters and thermal barrier coatings.
Thermal Stability and Oxygen Storage Mechanism
At the atomic level, cerium oxide’s thermal resistance comes from its fluorite crystal structure, which accommodates oxygen vacancies without collapsing. The ability to switch between Ce⁴⁺ and Ce³⁺ oxidation states allows it to release or absorb oxygen ions under varying temperatures.
This reversible oxygen storage capability not only stabilizes its structure but also enhances its performance in high-temperature reactions — a property that few other oxides possess.
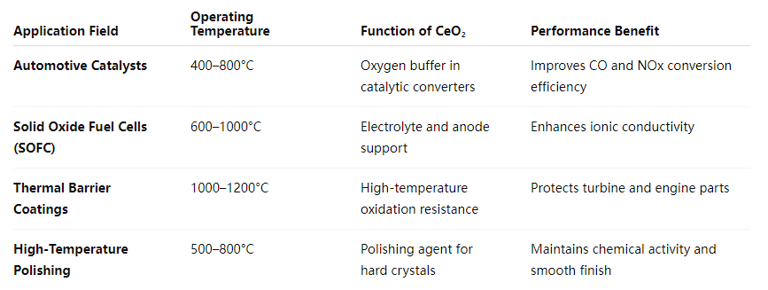
Industrial Applications at Elevated Temperatures
Doping and Nanostructuring: Enhancing CeO₂ Performance
To further improve cerium oxide’s performance under extreme conditions, scientists have explored doping with elements such as Zr, La, and Gd. These dopants enhance oxygen mobility and reduce sintering effects at high temperatures.
Meanwhile, nanostructured cerium oxide (nano-CeO₂) exhibits higher surface area and reactivity, enabling better catalytic efficiency and faster redox cycles.
Future Outlook: From Catalysis to Clean Energy
With the global push toward cleaner technologies, cerium oxide’s role is expanding beyond traditional catalysis. Its thermal endurance and reversible oxygen behavior make it promising for hydrogen production, CO₂ reduction, and next-generation energy systems. As research advances in sustainable synthesis and recycling, CeO₂ will continue to bridge the gap between material science and environmental innovation.
Conclusion
Cerium oxide’s success in high-temperature applications lies in the harmony between chemistry and stability. Its ability to endure, adapt, and react makes it one of the most versatile materials in modern industry — not only surviving the heat, but thriving in it.



Get in touch
Contact us to learn more about cerium oxide and its wide applications. Our team is ready to answer your questions and provide tailored solutions for your needs.
